 Antibodies labeled with small molecules such as fluorophore, biotin or drugs play a critical role in various areas of biological research,drug discovery and diagnostics. There are several limitations to current methods for labeling antibodies including the need for purified antibodies at high concentrations and multiple buffer exchange steps.
Antibodies labeled with small molecules such as fluorophore, biotin or drugs play a critical role in various areas of biological research,drug discovery and diagnostics. There are several limitations to current methods for labeling antibodies including the need for purified antibodies at high concentrations and multiple buffer exchange steps.
In a recent publication, a method (on-bead conjugation) is described that addresses these limitations by combining antibody purification and conjugation in a single workflow. This method uses high capacity-magnetic Protein A or Protein G beads to capture antibodies directly from cell media followed by conjugation with small molecules and elution of conjugated antibodies from the beads.
Using a variety of fluorophores the researchers show that the on-bead conjugation method is compatible with both thiol- and amine-based chemistry.
This method enables simple and rapid processing of multiple samples in parallel with high-efficiency antibody recovery. It is further shown that recovered antibodies are functional and compatible with downstream applications.
Literature Cited
Nidhi, N. et al. (2015) On-bead antibody-small molecule conjugation using high-capacity magnetic bead J. Immunol. Methods http://dx.doi.org/10.1016/j.jim.2015.08.008
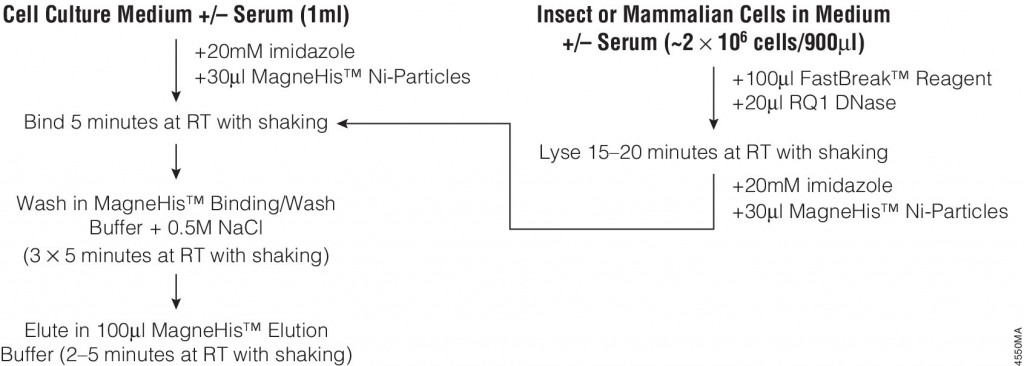
 Many different polypeptide fusion partners or affinity tags have been developed to facilitate purification of target proteins. The most commonly used tag for the purification and detection of recombinant expressed proteins is the His tag. Cloning vectors designed to generate His-tagged proteins contain 5–10 histidine residues at either the C- or N terminus of the expressed protein. The His tag adds only 0.84kDa to the mass of the protein and is nonimmunogenic. Also, because the tertiary structure of the tag is not important for purification, His-tagged proteins can be purified using native or denaturing conditions. The affinity of histidine residues for immobilized nickel allows selective purification of His-tagged proteins. The
Many different polypeptide fusion partners or affinity tags have been developed to facilitate purification of target proteins. The most commonly used tag for the purification and detection of recombinant expressed proteins is the His tag. Cloning vectors designed to generate His-tagged proteins contain 5–10 histidine residues at either the C- or N terminus of the expressed protein. The His tag adds only 0.84kDa to the mass of the protein and is nonimmunogenic. Also, because the tertiary structure of the tag is not important for purification, His-tagged proteins can be purified using native or denaturing conditions. The affinity of histidine residues for immobilized nickel allows selective purification of His-tagged proteins. The