Isolating DNA from plant tissues is difficult for many reasons. Unlike animal cells, plant cells have rigid cell walls, often made of tough fibrous material, and contain proteins and enzymes and other compounds such as polysaccharides and polyphenols that play a role in different cellular processes. These compounds can interfere with DNA isolation as well as downstream applications such as PCR. For these reasons, DNA isolation methods that are used successfully for other sample types may not work well to isolate DNA from plant material. Continue reading “DNA Purification from Plants: Not All Methods are Equal”
Isolating DNA from plant tissues is difficult for many reasons. Unlike animal cells, plant cells have rigid cell walls, often made of tough fibrous material, and contain proteins and enzymes and other compounds such as polysaccharides and polyphenols that play a role in different cellular processes. These compounds can interfere with DNA isolation as well as downstream applications such as PCR. For these reasons, DNA isolation methods that are used successfully for other sample types may not work well to isolate DNA from plant material. Continue reading “DNA Purification from Plants: Not All Methods are Equal”
#PromegaWorks
Descriptions of Promega products in action from examples of Promega products in the peer-reviewed literature to technical tips on how to address common laboratory and experimental questions
How Confident are You in Your Mass Spectrometry Data?
Data generated by scientific instruments and decisions based on that data depend on optimal instrument performance. Clinical assays rely on mass spectrometric (MS) data for accurate results so that correct health related results are gained and appropriate results-based decisions are made. However, there are no generally agreed upon tools nor performance standards for mass spectrometry. Furthermore, while several software tools exist that serve to assist with the analysis of instrument performance, a dedicated reagent software package has yet to be created. For optimal liquid chromatography (LC) performance, parameters like retention time, peak width and peak heightare typically reported. Commonly monitored MS parameters include mass accuracy, mass resolution, signal-to noise, sensitivity, limit of detection (LOD), limit of quantitation (LOQ) and dynamic range.
The 6 × 5 LC-MS/MS Peptide Reference Mix is designed for use in method development and optimization,and for routine liquid chromatography (LC) and mass spectrometry (MS) instrument performance monitoring. The product is a mixture of 30 peptides: 6 sets of 5 isotopologues of the same peptide sequence. The isotopologues (Figure 1) differ only by the number of stable, heavy-labeled amino acids incorporated into the sequence. The labeled amino acids consist of uniform 13C and 15N atoms. Each of the isotopologues is indistinguishable chemically and chromatographically. However, since they differ in mass, they are clearly resolved by mass spectrometry.
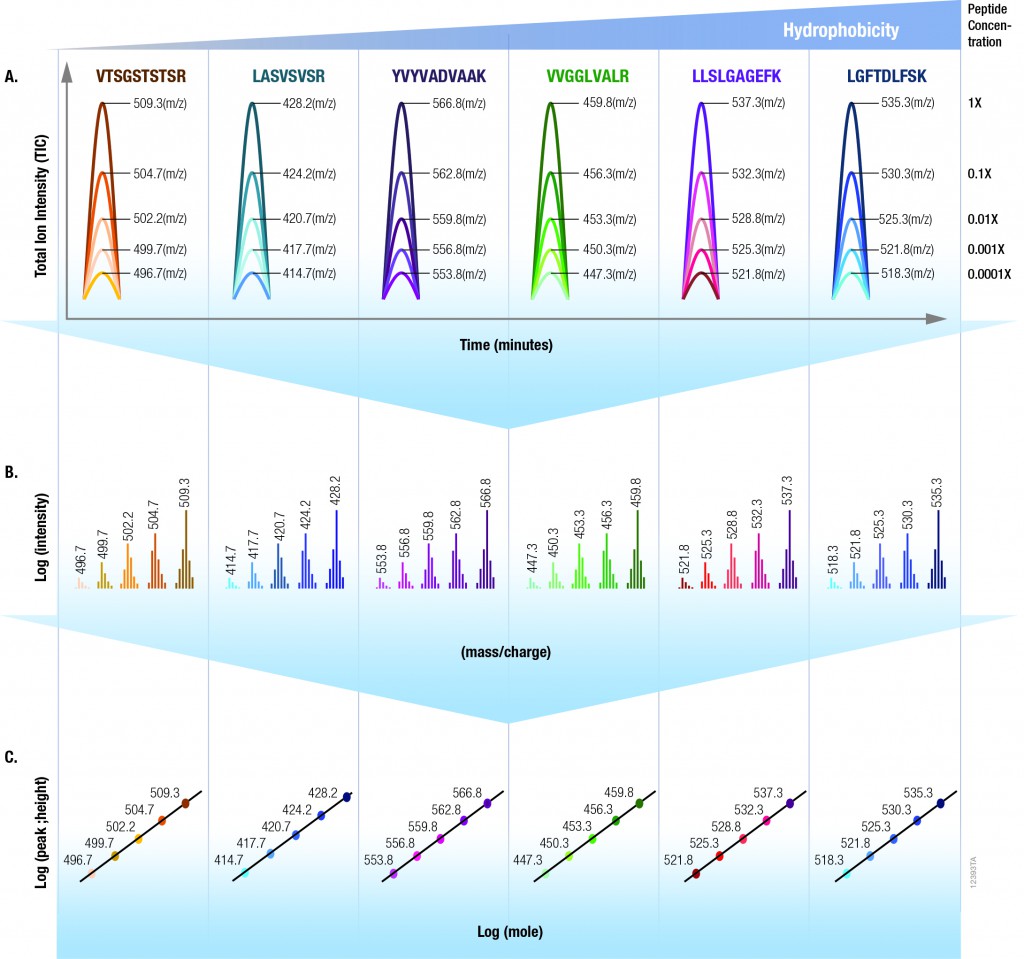
The isotopologues of each peptide are present in a series of tenfold differences in concentration or molar abundance. If 1pmol of the mixture is loaded onto an LC column, the next lighter isotopologue would be 100fmol, the next 10fmol, the second lightest 1fmol, and the lightest 100amol. This range allows assessment of the instrument’s dynamic range and sensitivity from a single run.
Peptides with a wide range of hydrophobicities were chosen to enable reporting of LC column performance. The most hydrophilic peptide gives users a tool to optimize the capture of hydrophilic peptides that might be difficult to capture otherwise, but that are too precious to use for method development.
To assist in data processing, a complementary software tool, is provided, the 6 × 5 LC-MS/MS Peptide Reference Mix Analysis Software (The PReMiS™ Software). The PReMiS™ Software produces a tabular report of calculated instrument parameters, graphical analysis of linearity curves as well as reporting the history of user-selected parameters such as LC retention time, peak height and mass accuracy. If the laboratory has a collection of instruments, there is also an option to compare parameters across instruments.
Detecting Inhibition of Protein Interactions in vivo
In a paper published in the September 2014 issue of ACS Medicinal Chemistry Letters, researchers from GlaxoSmithKline in the UK and Germany report on the discovery, binding mode and structure:activity relationship of a potent BRPF1 (bromodomain and PHD finger containing protein family) inhibitor. This paper came to our attention as it is one of the first publications to apply Promega NanoBRET technology in an vivo assay that reversibly measures the interaction of protein partners. The technology enabled the identification of a novel inhibitor compound that disrupts the chromatin binding of this relatively unstudied class of bromodomain proteins.
What exactly are bromodomains and why do they matter?
Bromodomains are regions (~100 amino acids) within chromatin regulator proteins that recognize and “read” acetylated lysine residues on histones. These acetylated lysines act as docking stations for regulatory protein complexes via binding of the bromodomain region. Because of their role in chromatin binding and gene regulation, bromodomains have attracted interest as potential targets for anti-cancer treatments. Although some bromodomain-containing proteins (e.g., those in the bromodomain and extraterminal domain (BET) subfamily) are well characterized and have been identified as potential therapeutic targets, others are less well understood.
Because Timing Is Everything….
 Timing is everything! I learned that the hard way just two weeks ago when I took my son to scout camp and thought I would try to capture the traditional American flag ceremony for posterity. I set up my camera for a panoramic shot and scanned the crowd. Feeling very pleased with myself, I got home that evening ready to show my family the great camera skills I had honed over the Summer months. To my horror, I noticed that half of the scout troop was saluting the flag while the other half were standing to attention! I had got the timing horribly wrong (although the picture is still fun to look at in a strange sort of way).
Timing is everything! I learned that the hard way just two weeks ago when I took my son to scout camp and thought I would try to capture the traditional American flag ceremony for posterity. I set up my camera for a panoramic shot and scanned the crowd. Feeling very pleased with myself, I got home that evening ready to show my family the great camera skills I had honed over the Summer months. To my horror, I noticed that half of the scout troop was saluting the flag while the other half were standing to attention! I had got the timing horribly wrong (although the picture is still fun to look at in a strange sort of way).
Timing is everything in science as well. As a technical services scientist at Promega I have sung the ‘timing’ tune to many a biologist. No more so than in the study of apoptosis where Caspases activate each other in a choreographed cascade of molecular triggers that all have their place and time in a domino sequence of enzymatic cleavage events. I frequently talk to researchers about that ‘sweet spot’ of activity when any given Caspase is busily cleaving a peptide moiety off of the next Caspase in the sequence. Finding that sweet spot is anything but trivial and often requires a considerable amount of patience during the optimization phase of experimentation.
Promega has developed a comprehensive suite of systems (see here) designed to help get the timing right for the cell and compound combinations you might be working with. The end result is that you have experiments that are timed so as to give you reliable information about what is really happening in your cells.
Freedom to Focus: Maxwell® Rapid Sample Concentrator

Back in the dark ages, when I was a graduate student, my idea of “automated” plasmid DNA extraction involved performing home-brew, “toothpick preps” in “strip tubes” or , if I was really feeling ambitious, a 96-well plate.
I would get just enough DNA to check for the presence of an insert, but the quality of the DNA was too low and the quantity too small to even consider using it for any other downstream experiments like amplification.
And increased throughput for other nucleic acid extraction needs? Nope. If I wanted genomic DNA, RNA or high-quality plasmid DNA, I spent time with columns and tubes, giving each sample my undivided individual attention.
Remember cesium chloride preps for RNA isolation? Even with the advent of column purification, which greatly simplified and standardized my protocols, nucleic acid purification was still a manual task that required a lot of time and effort to get the high-quality product I needed.
Doing the experiments that would answer the questions that I really wanted to ask (those “downstream experiments”), meant spending time at the bench performing careful (if tedious) work to isolate and clean up the highest quality nucleic acid possible. Even then inconsistency in sample prep could wreak havoc on downstream work.
Fortunately, for the modern scientist, personal, bench top automation, has progressed far beyond the toothpick and the strip tube to quality-tested, reliable nucleic acid extraction platforms like the Maxwell® Rapid Sample Concentrator (RSC).
The Maxwell® RSC improves sample preparation consistency, eliminating variability associated with manual handling, and your downstream results will reflect this consistency. With the RSC you can extract DNA or RNA from up to 16 samples in approximately 1 hour and viral total nucleic acids in less than an hour.
The instrument is easy to use: simply load the sample, push a button and walk away. Cross contamination is minimized and the instrument is supported by the Promega technical support and service you have come to trust over the past 35 years.
Want to know more about how the Maxwell® RSC can give you the freedom to focus on the work that interests you the most? To learn more, click here.
PowerQuant System: Tool for informed casework sample processing decisions
 Identification of a crime perpetrator on the basis of DNA fingerprinting is not as easy as some of the CSI shows on television make it out to be. A sample such as blood stain, touch sample or body fluid retrieved at a crime scene is often a challenge for DNA analysts. In many instances, the samples are limited in quantity, found in dirty conditions, exposed to harsh environmental factors and are mixtures of more than one DNA—human and/or non-human. One of the most important aspects of the workflow to successfully obtain a DNA fingerprint profile is accurate quantification of human amplifiable DNA. The more information gleaned from the sample, the better equipped the DNA analyst is to determine the best course of action for obtaining a usable short tandem repeat (STR) profile from challenging samples. Therefore, Promega has developed the PowerQuant™ System, a probe-based 4-target, 5-dye real-time PCR method to a) determine human and male DNA concentrations in a sample, b) detect possible PCR inhibitors c) identify possible mixtures and d) measure DNA integrity. Continue reading “PowerQuant System: Tool for informed casework sample processing decisions”
Identification of a crime perpetrator on the basis of DNA fingerprinting is not as easy as some of the CSI shows on television make it out to be. A sample such as blood stain, touch sample or body fluid retrieved at a crime scene is often a challenge for DNA analysts. In many instances, the samples are limited in quantity, found in dirty conditions, exposed to harsh environmental factors and are mixtures of more than one DNA—human and/or non-human. One of the most important aspects of the workflow to successfully obtain a DNA fingerprint profile is accurate quantification of human amplifiable DNA. The more information gleaned from the sample, the better equipped the DNA analyst is to determine the best course of action for obtaining a usable short tandem repeat (STR) profile from challenging samples. Therefore, Promega has developed the PowerQuant™ System, a probe-based 4-target, 5-dye real-time PCR method to a) determine human and male DNA concentrations in a sample, b) detect possible PCR inhibitors c) identify possible mixtures and d) measure DNA integrity. Continue reading “PowerQuant System: Tool for informed casework sample processing decisions”
For Alphavirus Reporters, Location Matters
Luminescent reporters offer virologists a convenient way to measure replication of viruses and are also used to image the spread of viruses in vivo in experimental systems. These reporter viruses are useful for evaluating the effects of antiviral drug treatments, testing the efficacy of potential vaccines, and studying the ways in which viruses replicate in the body and cause disease. One challenge in the construction of such reporters is the need to ensure that the reporter molecule itself does not alter the virus in ways that affect its ability to cause disease. Another challenge is maintaining the reporter gene throughout several cycles of viral replication. In smaller viruses, it can be particularly difficult to introduce a reporter gene without compromising the ability of the virus to replicate and cause disease.
A 2014 paper was published in J. Virology comparing the effectiveness of various NanoLuc® luciferase alphavirus reporter constructs. The authors of the study, Chengqun Sun et al. from the University of Pittsburgh, placed these reporter genes in three different locations in the genome of several alphaviruses and compared the effect on their ability to replicate in vitro and in vivo. They also assessed the ability of the luciferase genes to persist during infection of cultured cells and in a mouse model. They showed that the size and location of the reporter had a significant effect on successful replication and persistence. They also showed that the reporters could potentially be integrated at different positions within the genome to study different aspects of viral pathogenesis.
Continue reading “For Alphavirus Reporters, Location Matters”Novel Application for ProteaseMAX Surfactant: Cell Lysis
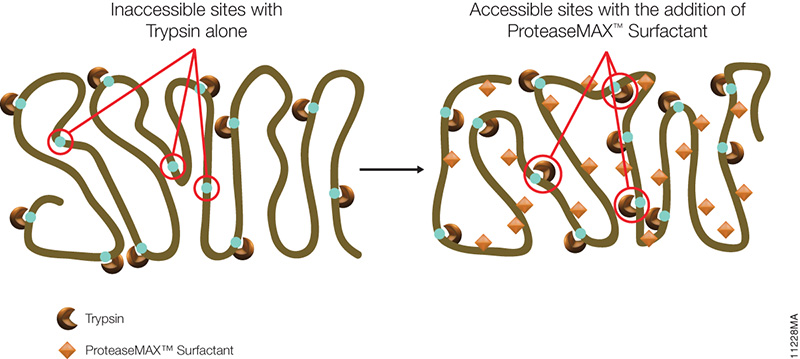
The novel mass spectrometry compatible surfactant sulfonate-(sodium 3-((1-(furan-2-yl)undecyloxy) carbonylamino)-propane-1-sulfonate (i.e.ProteaseMAX) facilitates both in-gel and in-solution digestion applications by reducing the time required, enabling protein solubilization/denaturation and increasing peptide/protein identifications.
A new application was highlighted in a recent publication (1) which utilized ProteaseMAX to lyse cells prior to trypsin digestion and subsequent mass spec analysis. The composition of the buffer determines the overall efficiency of cell lysis, dissociation of protein complexes, protein solubility and ease of removal prior to LC/MS-MS analysis.
Are you looking for proteases to use in your research?
Explore our portfolio of proteases today.
When compared to lysis buffers containing either urea or SDC, ProteaseMAX provided the optimal number of identified peptides/proteins.
In addition it can be easily removed from the lysate by acidic precipitation.
Reference
- Pirmoradian, M. et al. (2013). Rapid and deep human proteome analysis by single-dimension shotgun proteomics. Mol. Cell. Prot. 12, 3330–8.
Top 10 Innovator—Two Years Running
Antibody-dependent cell-mediated cytotoxicity (ADCC) is the main mechanism of action (MOA) of antibodies through which virus-infected or other diseased cells are targeted for destruction by components of the cell-mediated immune system. ADCC assays are often used to assess the effectiveness of monoclonal antibody therapies during the manufacture and development of biologic drugs. The bioluminescent ADCC Reporter Bioassays use an alternative readout at an earlier point in ADCC MOA pathway for the quantification of Fc effector function of antibody-based molecules: the activation of gene transcription through the NFAT (nuclear factor of activated T-cells) pathway in the effector cell.
The bioassay uses engineered Jurkat cells stably expressing the FcγRIIIa receptor, V158 (high affinity) variant, and an NFAT response element driving expression of firefly luciferase. The assay is ADCC MOA-based and features frozen, thaw-and-use effector cells and optimized reagents and protocol to perform a reporter-based ADCC bioassay in a single day. The ADCC Reporter Bioassay correlates with classic cytotoxic ADCC assays and is a suitable replacement for these cumbersome and highly variable assays.
The novel bioassay is linear, accurate, precise and stability indicating. Moreover, the bioassay shows good linear correlation between levels of glycosylation or fucosylation and ADCC activity. All of these features indicate the assay is suitable for use across biologic drug development programs.
Resources for the ADCC Reporter Bioassays:
-
Low Variability ADCC Bioassay
- ADCC Reporter Bioassay, Core Kit Technical Manual
- ADCC Reporter Bioassay, Complete Kit (WIL2-S) Technical Manual
- ADCC Reporter Bioassay Complete Kit (Raji) Technical Manual
- ADCC Reporter Bioassay Effector Cells, Propagation Model Technical Manual
- ADCC Reporter Bioassay: A Novel, Bioluminescent Cell-Based Assay for Quantifying Fc Effector Function of Antibodies (webinar)
NanoLuc® Luciferase: A Good Thing for Small Packages
A paper published on October 2 in the Journal of Virology describes an exciting development in the world of influenza research—the construction of a luciferase reporter virus that does not affect virulence and can be used to track development and spread of infection in mice.
Insertion of luciferase reporter genes into viruses has been accomplished before for several viruses, but has not been successful for influenza. Construction of influenza reporter viruses is complicated because the viral genome is small and all the viral genes are critical for infection. Therefore, replacement of an existing gene with a reporter gene or insertion of additional reporter sequences without affecting the virus’s ability to replicate and cause infection has proven difficult. To be successful, a reporter gene needs to be small enough to insert into the viral genome without eliminating any other vital functionality.
Continue reading “NanoLuc® Luciferase: A Good Thing for Small Packages”